- Article
- Source: Campus Sanofi
- 23 Oct 2023
An Introduction to Atopic Dermatitis
What is Atopic Dermatitis
Atopic dermatitis (AD) is a chronic disease driven by persistent underlying inflammation even in nonlesional or normal looking skin 1-3
- Atopic dermatitis is an immunological disease where the immune system causes more inflammation than normal
- The overactive immune system under the surface may lead to increased inflammation on the surface and is a contributing factor to the itchy patches on the skin
- Moderate-to-severe AD in adults is a chronic, debilitating inflammatory condition that requires long-term control 1,4
Challenges of treating Atopic Dermatitis
Atopic dermatitis (AD) is a chronic disease driven by persistent underlying Type 2 inflammation 1-3
Even skin that appears non-lesional on the surface is clearly distinct from normal skin:1-3
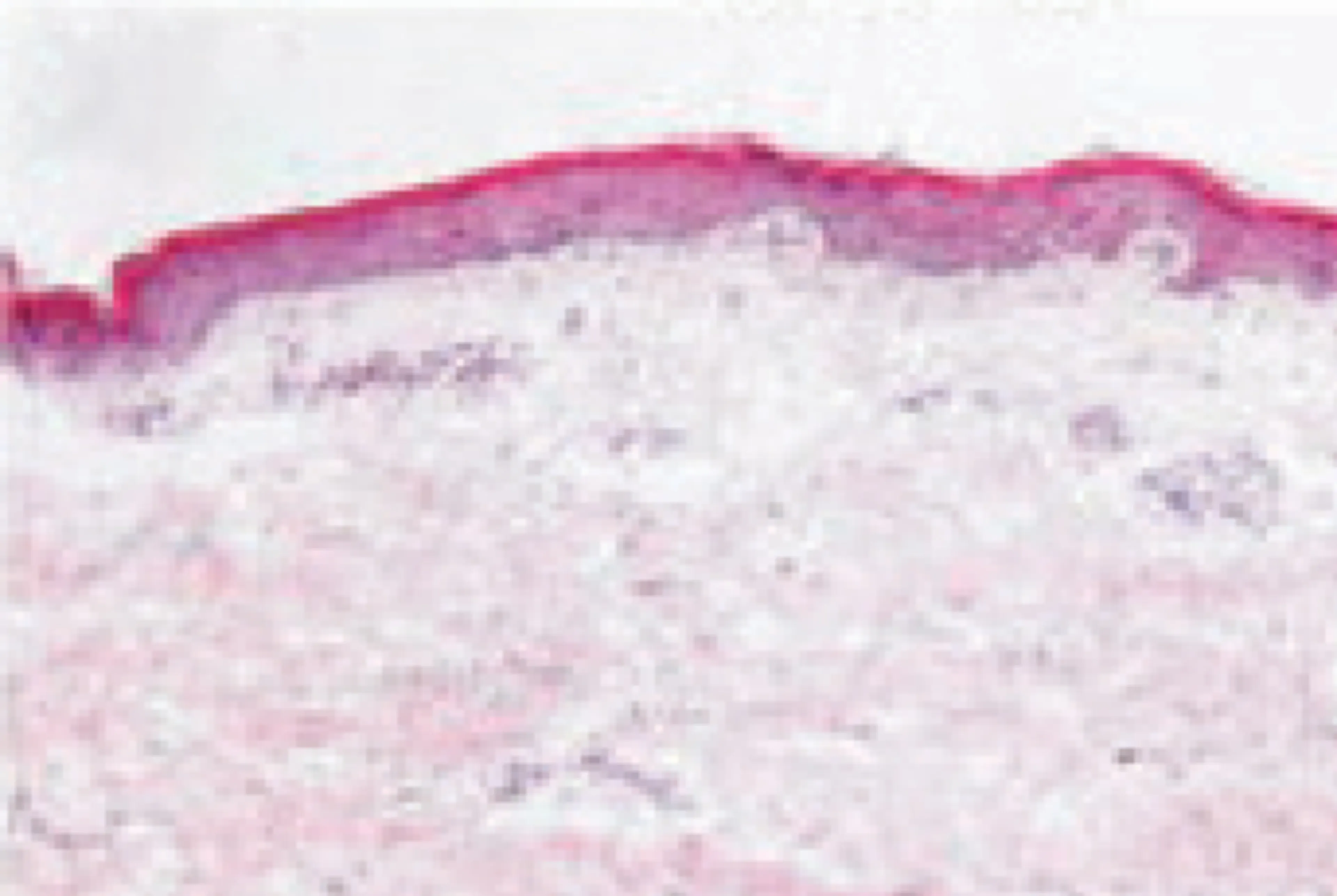
Normal skin

Lesional skin
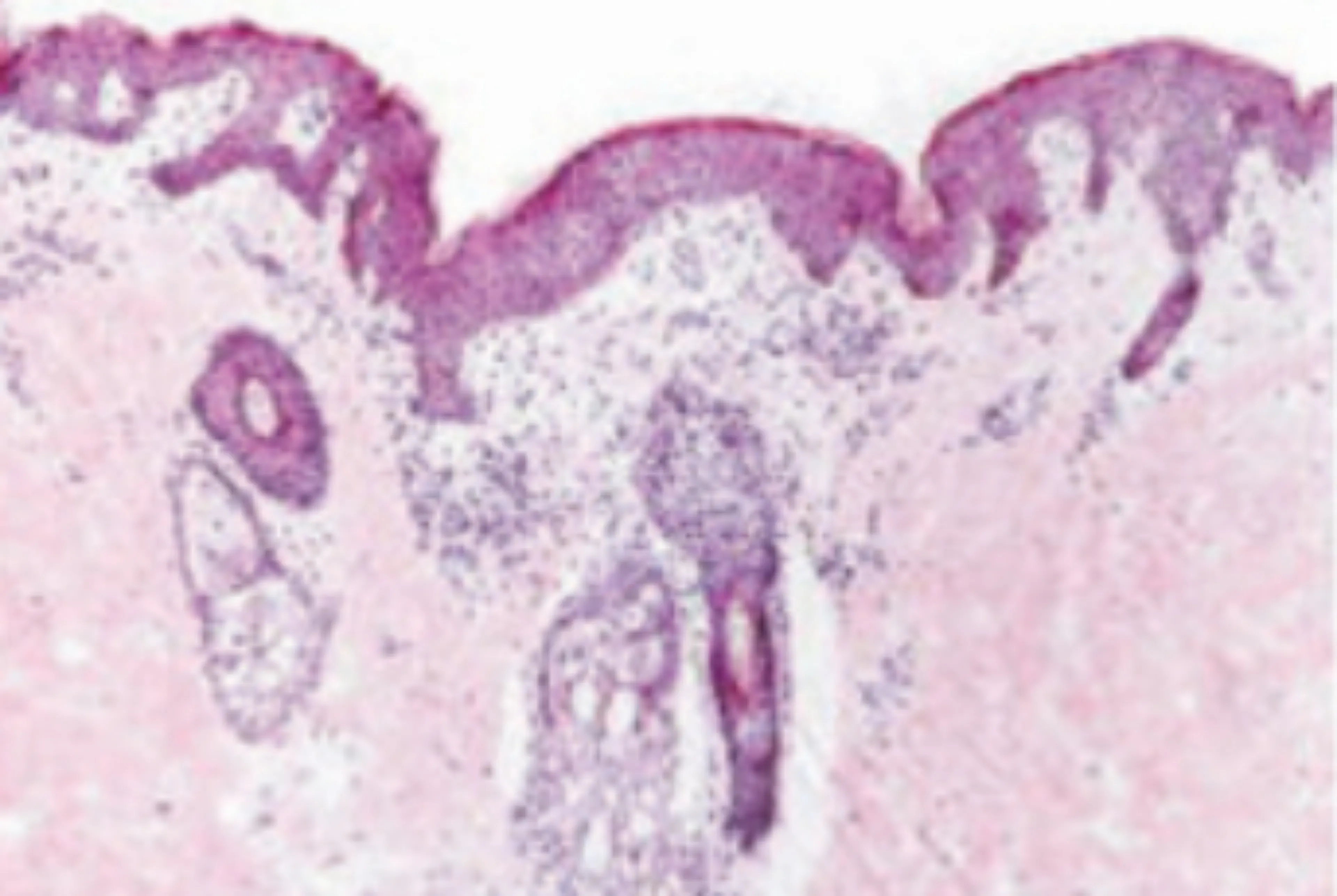
Non-lesional skin

Normal skin

Lesional skin

Non-lesional skin
Non-lesional skin in AD has been shown to have cutaneous expansion of T cells and abnormalities that are largely similar to lesional AD skin. This highlights the importance of early identification and treatment of patients with this condition.
- In the UK, ciclosporin is the only conventional systemic immunosuppressant approved for the management of severe AD in adults5. Other systemic immunosuppressants are used off-label, despite limited evidence supporting their efficacy and safety6-8
- Systemic immunosuppressants exert broad suppression of the immune system, while immunomodulators target specific immune pathways/cell types, thereby reducing off-target effects9
- Broad-acting immunosuppressants are associated with a range of toxicities and side effects that limit their long-term use and require routine laboratory monitoring to minimise the risks 6,7,10,11
Watch Dr. Woolf discuss the impact of immunosuppressant cycling:
AD experts* proposed four types of treatment failure which patients with AD may experience despite appropriate dose, duration and adherence to a therapeutic agent12:
- Inadequate clinical improvement12
- Failure to achieve stable long-terms disease control12
- Adverse events or poor tolerability with current treatment12
- Failure to relieve burden of disease† 12
*A steering committee that consisted of a multi-disciplinary group of AD experts, including 8 dermatologists, two allergists and a patient advocacy group representative. This group met to identify and prioritise a number of essential questions concerning the evaluation and management of moderate-to-severe AD in the era of biologic therapies
†Presence of ongoing impairment (e.g., pruritus, pain, loss of sleep, and poor quality of life) while on treatment
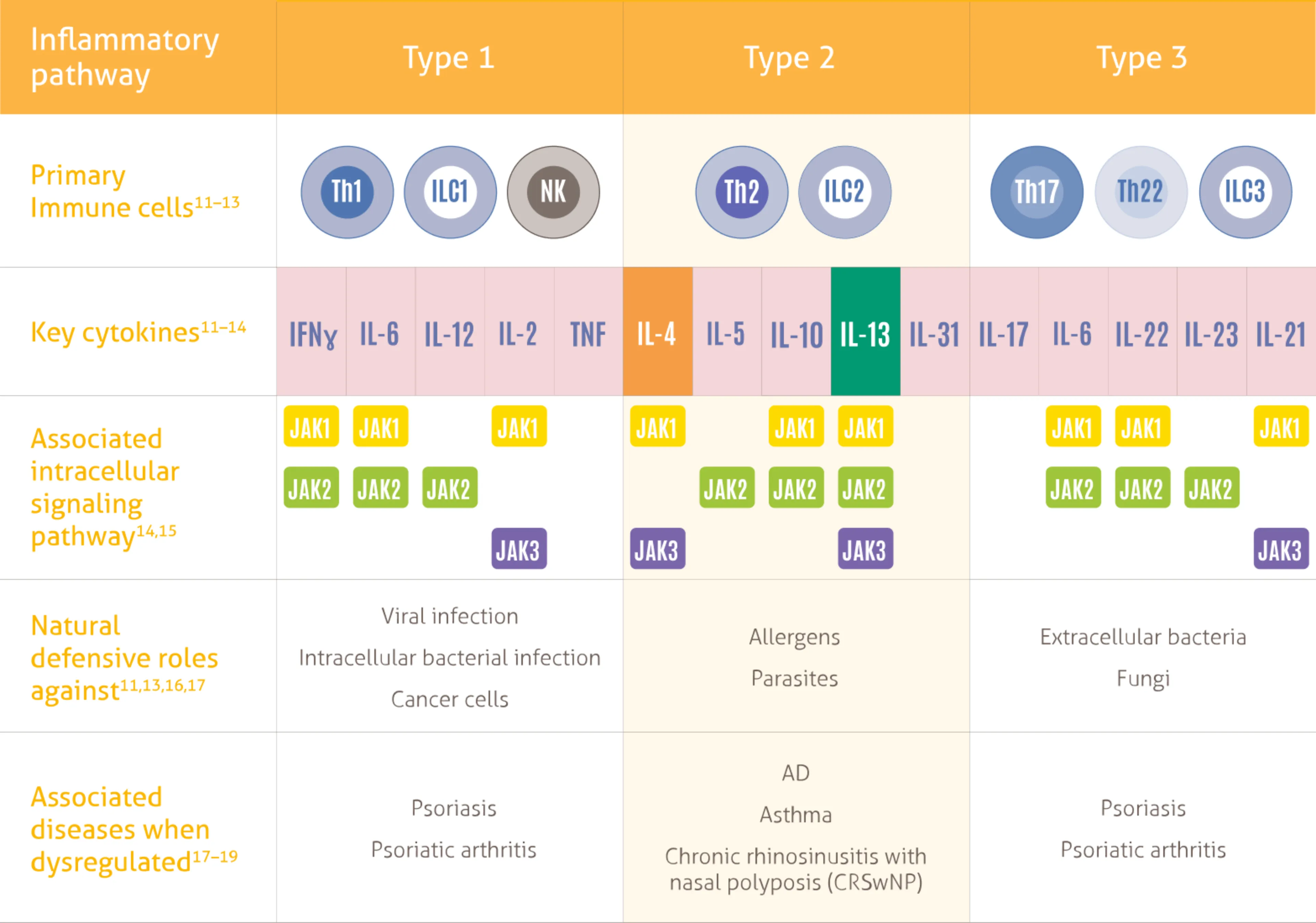
The Role of Type 2 Inflammation
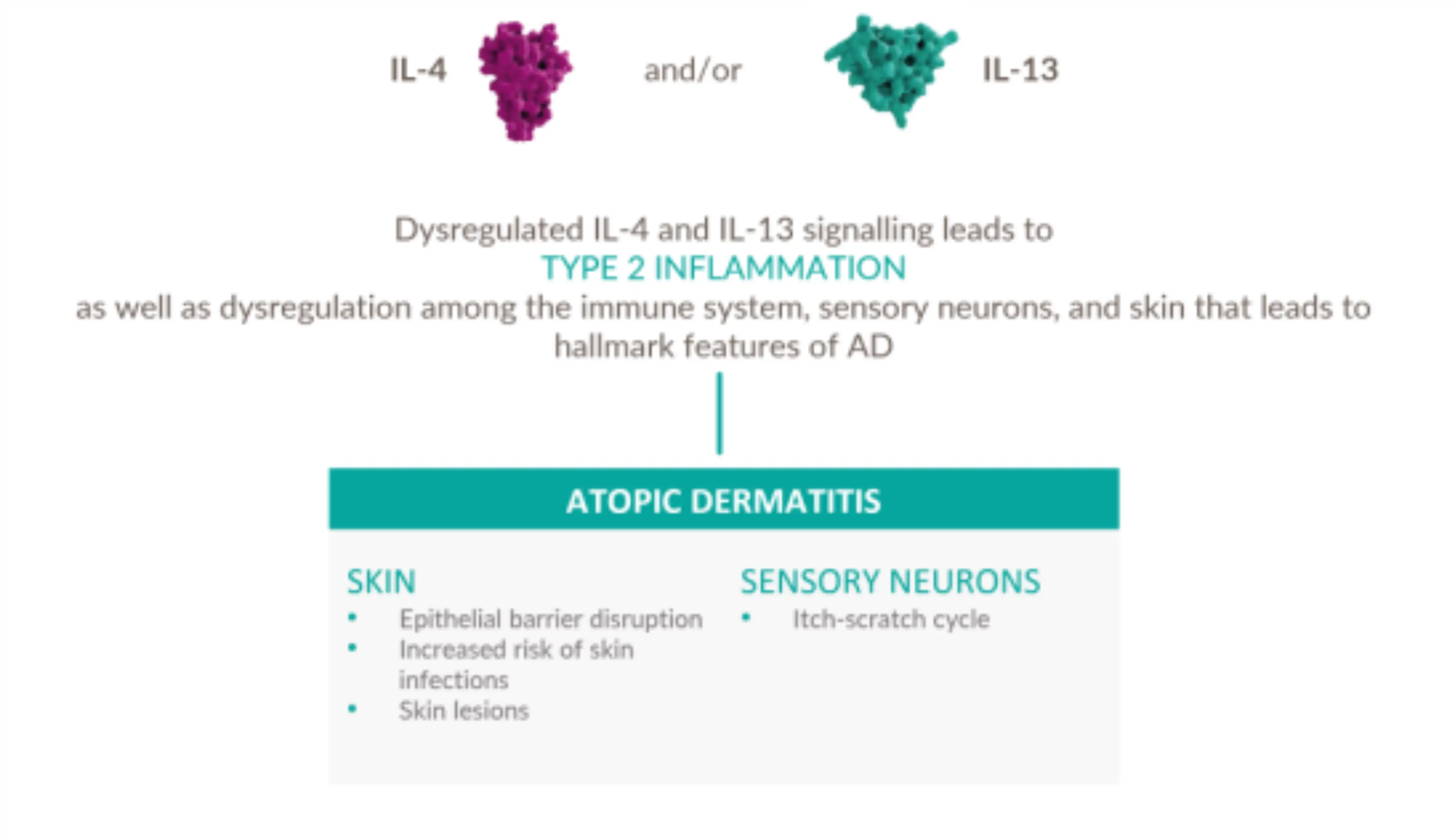
Dysregulated signaling of IL-4 and IL-13—two key drivers of type 2 inflammation—plays an important role in the pathogenesis of AD 13-18
IL-4 is a key upstream modulator of Th2 response, promoting a positive feedback loop that contributes to dysregulated secretion of IL-4, IL-13, and IL-31 #
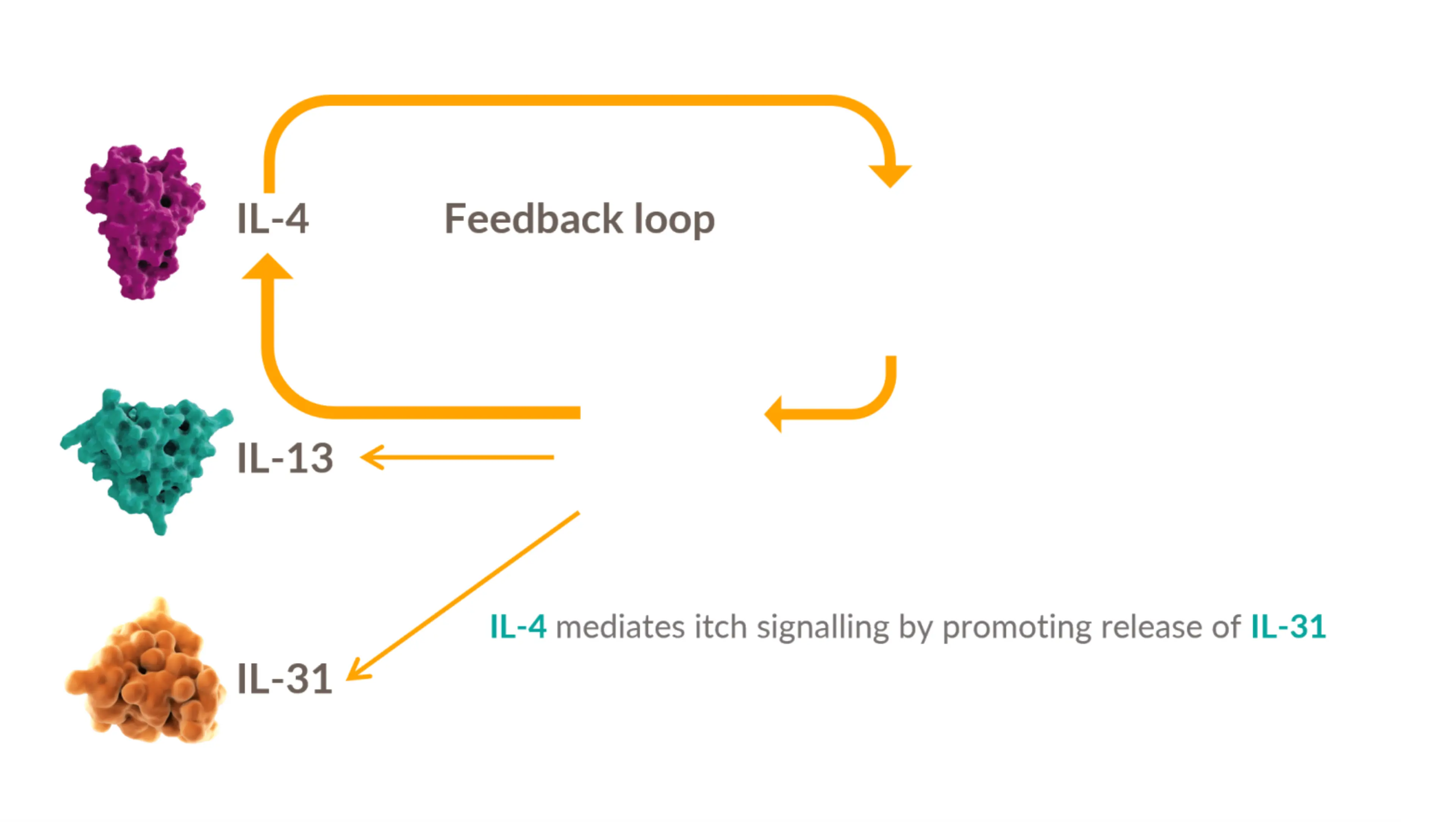
IL-4 and IL-13 have overlapping roles in Type 2 inflammation:
Watch Dr Alan Irvine discuss the role of IL-4 in Type 2 Inflammation for more information
Management approaches in AD should address the persistent underlying Type 2 inflammation
- Three types of immune response play a natural protective role against different pathogens, the Type 2 immune response drives AD specifically 1-3
- Broad spectrum immunosuppressants do not specifically inhibit Type 2 inflammatory pathway 19-21
- Non-specific mechanisms may result in unwanted side effects and the need for lab monitoring, limiting traditional systemic treatments as long-term therapeutic options 1,19-24
Watch this video to hear Dr Woolf discussing the immunological targets at play in AD:
DUPIXENT directly targets the underlying type 2 inflammation involved in AD.
References
- Leung DYM, et al. J Clin Invest. 2004;113(5):651–657.
- Suárez-Fariñas M, et al. J Allergy Clin Immunol. 2011;127(4):954–964
- Gittler JK, et al. J Allergy Clin Immunol. 2012;130(6):1344–1354.
- Huang A, et al. Curr Treat Options Allergy. 2017;4(3):355–369
- Neoral Soft Gelatin Capsules. Summary of Product Characteristics. Available at https://www.medicines.org.uk/emc/product/1034/smpc
- Wollenberg A, et al. J Eur Acad Dermatol Venereol. 2018;32(6):850-878
- Wollenberg A, et al. J Eur Acad Dermatol Venereol. 2022;36(9):1409-1431
- Bieber T, Straeter B. Allergy. 2015;70:6–11.
- Janssen M, et al. Intensive Care Med. 2023;49(4):462-4
- Berth-Jones J, et al. Br J Dermatol 2019;180(6):1312–38
- Armstrong AW, et al. PLoS ONE. 2019;14(1):e0210517
- Boguniewicz M, et al. J Allergy Clin Immunol Pract. 2017;5:1519–1531.
- DUPIXENT Summary of Product Characteristics. 2023
- Serezani APM et al. J Allergy Clin Immunol 2017;139(1):142-151.
- Silverberg JI et al. Dermatol Clin 2017;35(3):327-334
- Nguyen JK et al. Arch Dermatol Res 2020;312(2):81-92.
- Haddad E-B et al. Dermatol Ther (Heidelb) 2022;12(7):1501-1533
- Oetjen LK et al. Cell 2017;171(1):217-228
- Hajar T, et al. An Bras Dermatol. 2018;93:104–107
- Wakelin SH, et al. Medicine. 2017;45:363–367
- Moyle M, et al. Exp Dermatol. 2019;28:756–768
- Seegräber M, et al. Expert Rev Clin Pharmacol. 2018; 11:467–474
- Armstrong AW, et al. PLoS ONE. 2019:14:e0210517
- Wu J et al. Expert Opin Biol Ther. 2020;20:525–538
- Kaiko GE, et al. Immunol. 2008;123:326–338.
- Eyerich K et al. J Eur Acad. 2017;32:692–703.
- Raphael I, et al. Cytokine. 2015;74:5–17
- Schwartz DM, et al. Nat Rev Rheumatol. 2016;12(1):25–36
- Murray PJ. J Immunol. 2007;178(5):2623–2629
- Annunziato F, et al. J Allergy Clin Immunol. 2015;135(3):626–635
- Schleimer R. Annu Rev Pathol. 2017;12:331–357
- Nakayama T, et al. Annu Rev Immunol. 2017;35:53–84.
- Coates LC, et al. Semin Arthritis Rheum. 2016;46:291–304

Atopic Dermatitis Control Test (ADCT)
Atopic dermatitis, a type of eczema, may be affecting your patient’s life in more ways than you know.
The ADCT gives a measure of how controlled your patient’s eczema is. Use these 6 concise questions to evaluate all dimensions of atopic dermatitis control.
Try the ADCTMAT-XU-2304408 (v1.0) Date of Preparation October 2023